AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
Back to Blog
Covid rash vaccine9/25/2023  These usually transient AEs are very frequently reported after COVID‐19 vaccinations as well. 1, 2, 3, 4, 5 General symptoms that have also frequently been observed after the use of other vaccines include fever, headache, fatigue, chills, muscle pain, diarrhoea and local injection‐site reactions. Hence, there is also increasing real‐world evidence on adverse events associated with the use of the new COVID‐19 vaccines. End of May 2021, over 1.5 billion vaccine doses have been administered worldwide. Most vaccines need two doses, except for Janssen COVID‐19 vaccine ® and Convidecia ®. All these vaccines have to be administered by intramuscular injection. 1, 2, 3, 4, 5 The vaccine platform of aforementioned vaccines includes mRNA, non‐replicating virus vector or inactivated virus material. However, there are other COVID‐19 vaccines, including Convidecia ® (CanSino Biologics), Sputnik V ® (Gamaleya Research Institute), CoronaVac ® (Sinovac), which have been approved by at least one country. Moreover, the vector‐based vaccine Janssen COVID‐19 vaccine ® (Johnson & Johnson ) has now also been approved for use in Europe. Since December 2020, huge vaccination campaigns have been introduced in Europe, using first the mRNA vaccines Comirnaty ® (Pfizer/BioNTech BNT162b2) and Moderna ® (Moderna mRNA‐1273) and then also the viral vector‐based vaccine Vaxzevira ® (AstraZeneca AZD1222) which all have recently been approved by the European Medicines Agency. 1, 2, 3, 4, 5 According to the WHO COVID‐19 dashboard accessed on 1 June 2021, there have been more than 170 million confirmed cases of COVID‐19, including almost 3.6 million deaths. However, SARS‐CoV‐2‐specific and neutralizing antibodies decrease over about 8 months, raising concerns for long‐term protection and herd immunity post infection. Memory B cell as well as CD4+ lymphocyte responses can be detected in over 90% of patients up to 6 months after infection. A significant variability in the magnitude of adaptive immune responses has been observed in patients with COVID‐19. Apart from antibody formation, natural infection with SARS‐CoV‐2 also activates a T helper lymphocyte‐1 pronounced immune response with SP‐specific CD4+ lymphocytes. Hence, neutralizing antibodies are probably the main players in protection from the virus. Antibodies particularly binding to the receptor‐binding domain and the N‐terminal domain of SP can prevent virus attachment to cells finally resulting in neutralization of the pathogen. S1 includes the receptor‐binding domain finally binding to the ACE‐2 receptor. 
Subunits (S1, S2) of the trimeric SP are involved in receptor binding as well as membrane fusion activity. Via the SARS‐CoV‐2 spike protein (SP) at the virus surface, the pathogen is capable to enter human cells by binding to the angiotensin‐converting enzyme‐2 (ACE‐2). As a result of the very fast spread of the disease throughout the globe, the World Health Organization (WHO) declared COVID‐19 a pandemic on March 2020. In the end of December 2019, a novel coronavirus disease (COVID‐19) caused by the severe acute respiratory syndrome coronavirus 2 (SARS‐CoV‐2) emerged in China.  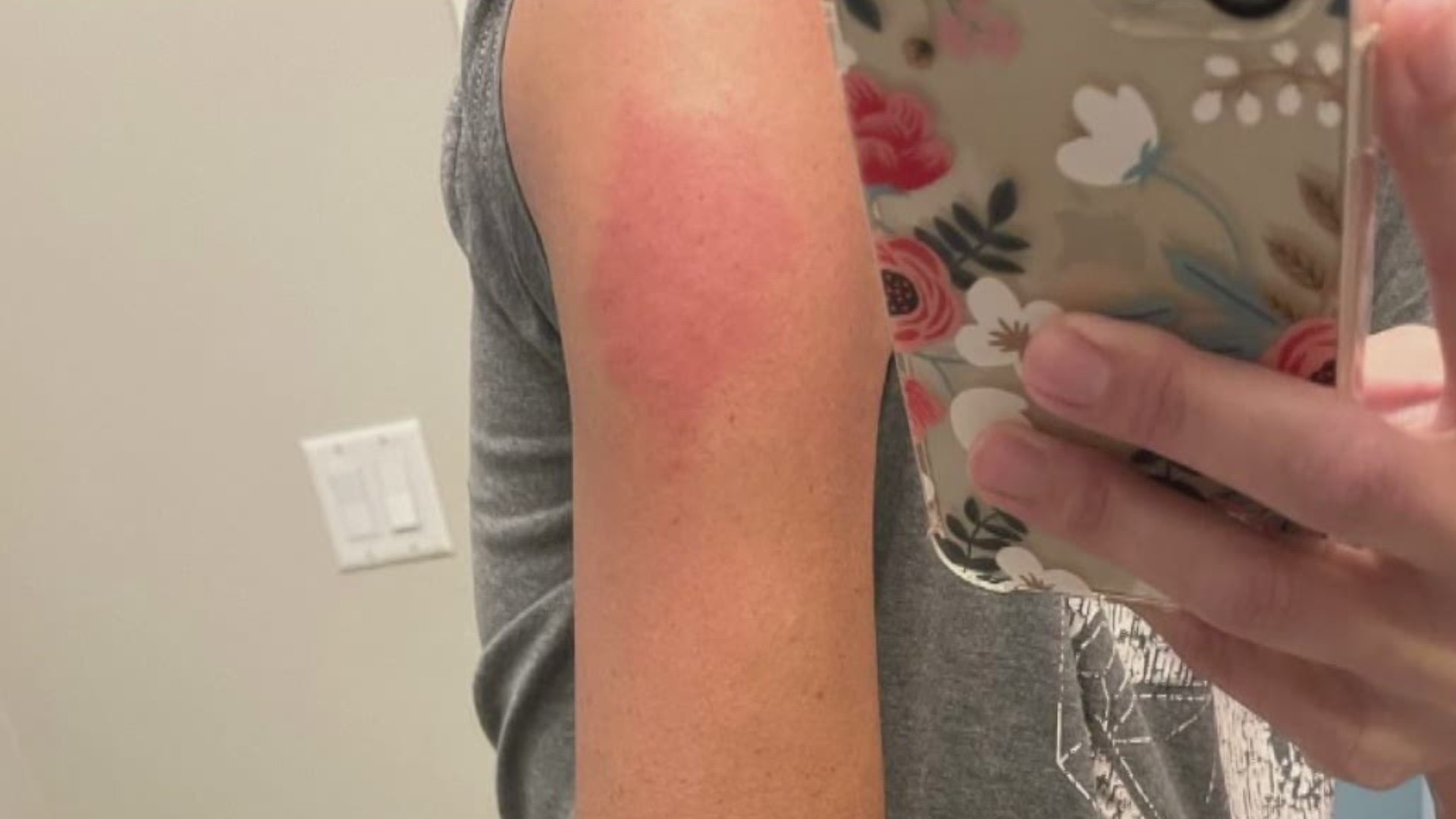
the spike‐protein sequences used to design the vaccines) and human components and may thus explain some COVID‐19 pathologies as well as adverse skin reactions to COVID‐19 vaccinations. Importantly, molecular mimicry exists between SARS‐CoV‐2 (e.g. In conclusion, there are numerous cutaneous reaction patterns that may occur following COVID‐19 vaccination, whereby many of these skin findings are of immunological/autoimmunological nature. Pityriasis rosea‐like rashes and reactivation of herpes zoster have also been reported after COVID‐19 vaccination. Functional angiopathies (chilblain‐like lesions, erythromelalgia) may also be observed. Autoimmune‐mediated skin findings after COVID‐19 vaccination include leucocytoclastic vasculitis, lupus erythematosus and immune thrombocytopenia. Type IV hypersensitivity reactions may be observed, including delayed large local skin lesions (“COVID arm”), inflammatory reactions in dermal filler or previous radiation sites or even old BCG scars, and more commonly morbilliform and erythema multiforme‐like rashes. urticaria, angio‐oedema and anaphylaxis) likely due to allergy to ingredients may rarely occur but can be severe. Unspecific injection‐site reactions very early occurring after the vaccination are most frequent. In this review article, we provide a brief overview on cutaneous findings that have been observed since the emerging mass COVID‐19 vaccination campaigns all over the world. 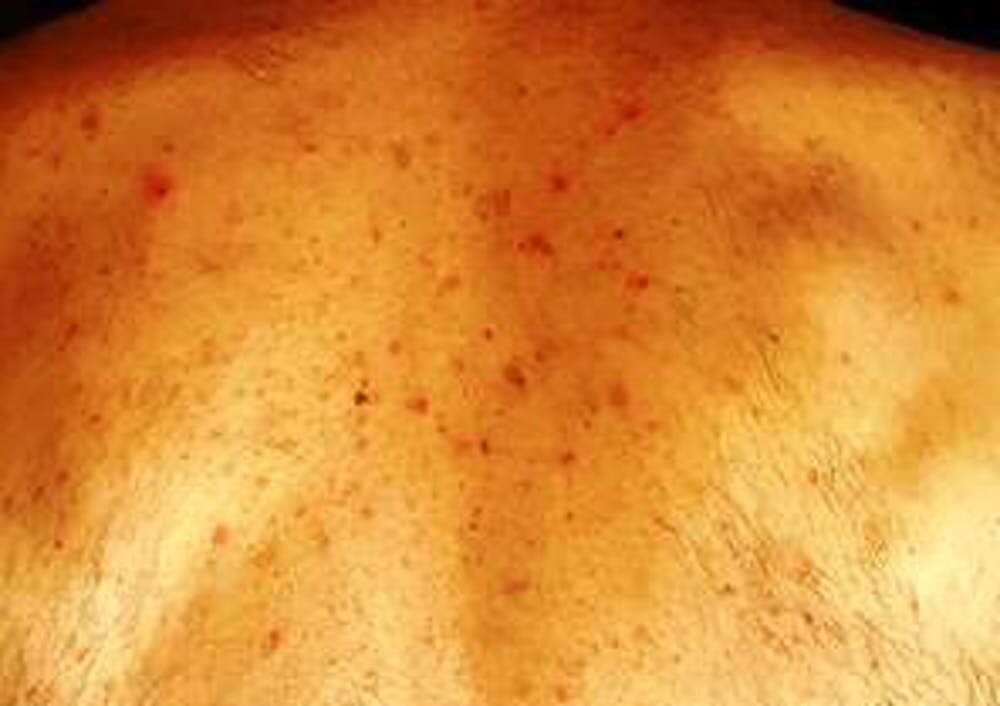
There is growing evidence that not only the novel coronavirus disease (COVID‐19) but also the COVID‐19 vaccines can cause a variety of skin reactions. 
0 Comments
Read More
Leave a Reply. |
 RSS Feed
RSS Feed
